Energy storage and sustainability
The development and implementation of renewable energies generated by wind, solar or tidal powers are increasing and set an important milestone for the change towards a sustainable energy society. However, the revolution of the energy landscape is only possible with the implementation of stationary and mobile energy storage technologies able to balance energy fluctuations or use on demand.
The growing ratio of electro-mobility and consumer electronics, including smartphones, tablets and laptops, requires mobile high-performance energy storage technologies like lithium-ion batteries. High-performance energy storage devices are characterised by a high specific energy and power (energy per weight) (Figure 1). In other words, electric vehicles with a high specific energy can drive longer distances. In addition, a high specific power means that the electric vehicle can accelerate very fast. However, the monopoly of lithium-based batteries has one major issue: sustainability. Flammability, difficulty for recycling and limited availability of raw materials raise the question: How can we develop high performance batteries with safe cell reactions, earth-abundant materials and long shelf life?
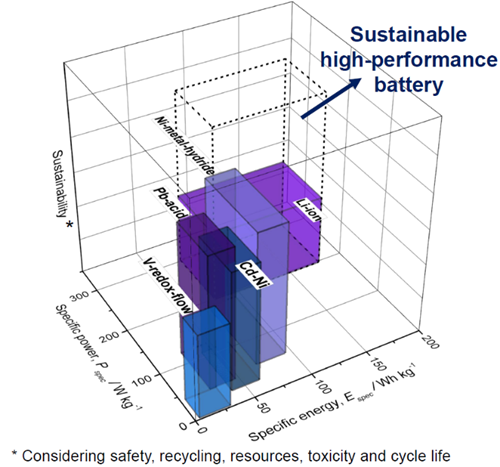
Figure 1: Specific power and energy in relation to sustainability of state-of-the-art rechargeable battery systems.
Alternative batteries are the new trend
There is a strong an increasing trend in the battery community towards alternative battery systems based on ready available and abundant elements like sodium, magnesium or aluminium in order to form a variety of battery systems, which balance the demand of lithium. The proposed battery systems should show a high potential of sustainability in terms of availability of resources, safe cell reactions, cost and theoretical performance. However, it needs to be noticed that sustainability is a relative term. An alternative battery system cannot be 100% recyclable, cost-effective and environmentally friendly, but more sustainable in comparison to other systems, like lithium-ion batteries.
The current state of performance of these alternative battery systems cannot yet compete with existing lithium-based batteries. In order to see the bigger picture, it merits investigating towards alternative systems. Looking back approximately 60 years ago, fossil oil was equated with profusion, technological progress and expansion. The same image applies for lithium-based technologies today. However, it is doubtful if the lithium resources can catch up economically with the lithium demand in the next 50 years, paraphrasing, lithium might be the next oil.
Therefore, the balance between performance and sustainability of battery systems became a key challenge. In order to overcome this obstacle, new energy storage technologies need to be proposed or already existing technologies need to be combined in a novel approach.
Combination of battery and capacitor
The hybrid battery-capacitor In contrast to capacitors, batteries are limited to reach a high specific power. Unlike like batteries, which are chemical energy storage devices, capacitors store energy physically. This allows fast charge and discharge processes with high power output. High-performance batteries are characterised by their high energy per mass, based on long discharge times and voltages.
A promising approach to combine the advantages of a battery and capacitor in one system was proposed by a joint project of the University of Southampton (UK), Technische Universität Ilmenau (Germany) and Hokkaido University (Japan) sponsored by the International Consortium of Nanotechnology of the Lloyds Register Foundation (ICON-LRF) and Engineering and Physical Sciences Research Council (EPSRC).
The proposed battery system based on metallic aluminium at the negative electrode and a conductive polymer at the positive electrode in an ionic liquid electrolyte. The battery components are earth-abundant, good recyclable and safe.
The specific feature of this energy storage device is the combination of a battery oxidation-reduction-reactions at the positive and negative electrodes, respectively and at the same time the storage of electrical charges as it is typical for a capacitor. When the battery is charged, at the negative electrode metallic aluminium is electrochemically deposited (reduction reaction) and at the positive electrode the conductive polymer is charged from neutral to a conductive state (oxidation reaction). The special characteristic is that at the positive electrode, anions of the ionic liquid electrolyte are intercalated simultaneously into the nano-structured pores of the polymer backbone and stored as an electrical charge like in a capacitor. When the battery is discharged, the opposite reactions occur, aluminium dissolves (oxidation reaction), and the conductive polymer changes to a neutral charged state (reduction reaction) and de-intercalates the anions from the polymer.
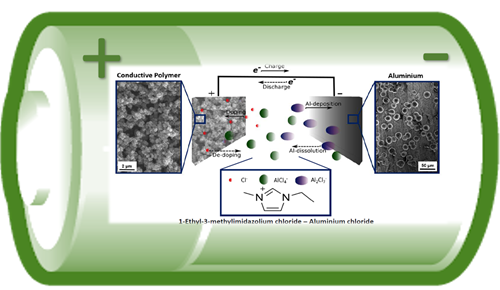
Figure 2: Schematic sketch the charge and discharge reaction in a conductive polymer-aluminium hybrid battery-capacitor.
There are promising preliminary performance parameters of the proposed hybrid- system that are in the range of the state of the art nickel-metal-hydride and lithium-ion batteries. However, the system had so far one limitation, which was the stability of the conductive polymer electrode. This influenced the life-time and caused a fast failure after a few charge-discharge cycles of the battery system.
The reason for the electrode instability was investigated and remedied by synthesising conductive polymers in ionic liquids, which are similar to the used electrolyte in the battery system. Furthermore, the design of the conductive polymer electrode was reconsidered. Instead of a one-dimensional electrode surface, a 3D with structures in the micro- and nano scale pledges higher performances and stability (Figure 3).
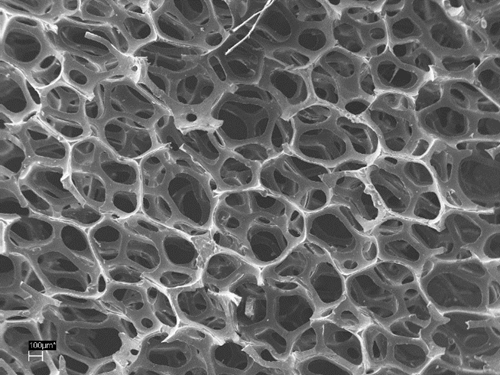
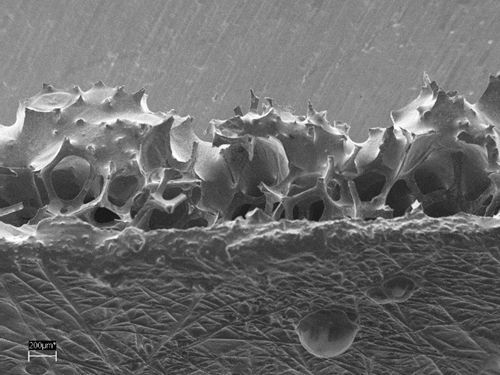
Figure 3: Scanning electron microscope images of 3D conductive polymer-carbon composites.
Back to the fundamentals
The current research regarding the described system took a step back. In order to improve the performance of the hybrid-system, the fundamental electrode reactions and mechanism at the interface electrode-electrolyte must be understood. The reactions of the metal electrode, the deposition and dissolution of aluminium in the ionic liquid electrolyte, is extensively studied and understood well. However, the performance of the hybrid-system is mainly determined by the conductive polymer electrode, which still holds several open questions. The main aspects that require responses are:
Further studies are required to answer these questions and proceed with the applied research of the hybrid-system.
At the moment, the teams in the UK and Germany are investigating the doping and de-doping mechanism of the conductive polymers in ionic liquids by quartz crystal microbalance measurements, a powerful method to study the active doping species into the conductive polymer as well as the reversibility of the system. Furthermore, these measurements will be accompanied by in-operando atomic force microscopy investigations in order to understand the structural changes suffered by the conductive polymer during the battery charging and discharging cycles.
These fundamental but important investigations will form the basis of the functional understanding of the hybrid-battery-capacitor system and will allow transferring the gained models on other similar energy storage systems, which will contribute to a more sustainable energy future.