Sunlight is arguably the most abundant clean source of energy that is capable of enabling “indefinite” and sustainable economic growth, with minimum detrimental impact on the environment. Crystalline silicon solar cells have been continuously advancing in efficiency and reducing in cost of fabrication over the last 40 years, and in some locations in the world at the current prices, they are capable of producing electricity from sunlight at a comparable cost to the price of electricity generated from fossil fuels.

Solar Cells
There are many other newer solar cell technologies that promise even lower cost solar power, and these range from thin-film vapour deposited semiconductor-based solar cells to solution-processed solar cells based on organic semiconductors, hybrid composites, or inorganic semiconductors, often referred to as second- and third-generation photovoltaics (PVs), respectively. The third generation, or “emerging technologies”, has attracted a massive increase in research effort over the last 10 years, which has led to tremendous advances in efficiency, reproducibility, and stability, and the first commercial products are being sold for niche markets to power consumer electronics and low-power applications in buildings.
However, to compete for the larger power markets and indeed drive the adoption of solar cells for utility-scale power generation, a significant increase in efficiency is required for these emerging technologies. Stemming from research in solution-processable semiconductors, a relatively old family of materials has emerged as a serious contender for utility-scale solar power. Solar cells based on perovskite absorbers promise to break the prevailing paradigm by combining both ultimately low cost and high efficiency.
“Perovskites” is the nomenclature for any materials that adopt the same crystal structure as calcium titanate. There are hundreds of different materials that adopt this structure, with a multitude of properties. The general chemical formula for perovskite compounds is ABX3, where 'A' and 'B' are two cations of very different sizes, and X is an anion that bonds to both. The 'A' atoms are larger than the 'B' atoms.
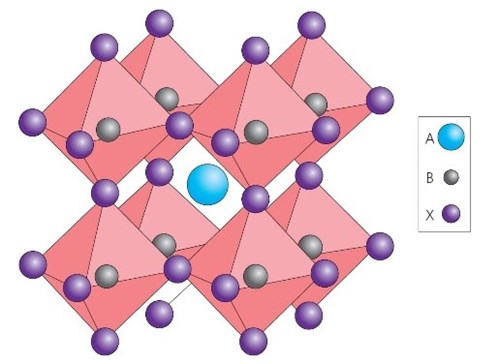
Perovskite crystal structure
Synthetic perovskites have been identified as possible inexpensive base materials for high-efficiency commercial photovoltaics. They showed a conversion efficiency of up to 20%. Methylammonium tin halides and methylammonium lead halides are of interest for use in solar cells. Due to their high photoluminesence efficiencies, perovskites are also good candidates for use in light-emitting diodes.

Perovskite solar cells
Nanometer-scale crystals [nanocrystals (NCs)] represent the most recent type of lead halide perovskites materials with promising applications. Recent studies have shown that these materials could deliver high efficiency at any desired wavelength in the visible spectrum. Even though research on perovskite nanocrystals is still at the starting stage, they are expected to have a significant impact in the development of novel lighting devices in the near future thanks to their facile synthesis compared to other semiconductor nanocrystals.