Transition metal oxides such as titanium dioxide (TiO2) are considered as an extremely important class of materials due to their intense catalytic activity, superior chemical stability, long life cycle and high electrical conductivity. Particularly, anatase TiO2 has been extensively studied because of its abundance and non-toxicity, high photocatalytic activity and responseto visible light arising from high its absorption coefficient and reflectivity. Moreover, it represents the work-horse photoanode material in dye-sensitized solar cells (DSSCs) whereas recently has been widely applied as the device electron transport material in both organic-inorganic halide perovskite (PSC) and organic solar (OSC) cells. Nevertheless, TiO2 exhibits a wide band gap of around 3.2 eV that limits its absorption in the visible and, especially, in the near infrared (NIR) region. A common way to reduce the band gap of TiO2 is through doping with appropriate elements which has also significant impact on its electronic structure. A vast variety of literature reports previously demonstrated that by doping TiO2 with nitrogen (N), halogens such as fluorine (F) and chlorine (Cl), or several transition metal ions such as zinc (Zn) or nickel (Ni), significant changes in its electronic structure are observed. Particularly, the formation of mid gap states resulting in a band gap reduction was evident. For instance, when titanium dioxide is doped with Ni the band gap decreases to 2.57 eV, whereas when it is doped with Cl mid gap states are formed and the band gap decreases to 3 eV. Progress on the photocatalytic performance and heterogeneous catalysis of TiO2 and other oxides such as ZnO has also been accomplished by the introduction of oxygen vacancies combined with metallic doping.
Focusing on other defect related projects regarding the TiO2 and its application to photocatalysis, it is seen that TiO2 has a fast recombination of the conduction band electrons and valence band holes and as a result it is not a satisfactory photocatalytic for organic degradation. In order to solve that problem as well as to reduce the large band gap, it is seen in the literature that many doped models of TiO2 can have improved photocatalysis. For example, N doped TiO2 and Nb doped TiO2 is reported to have better photocatalytic properties than pure TiO2.
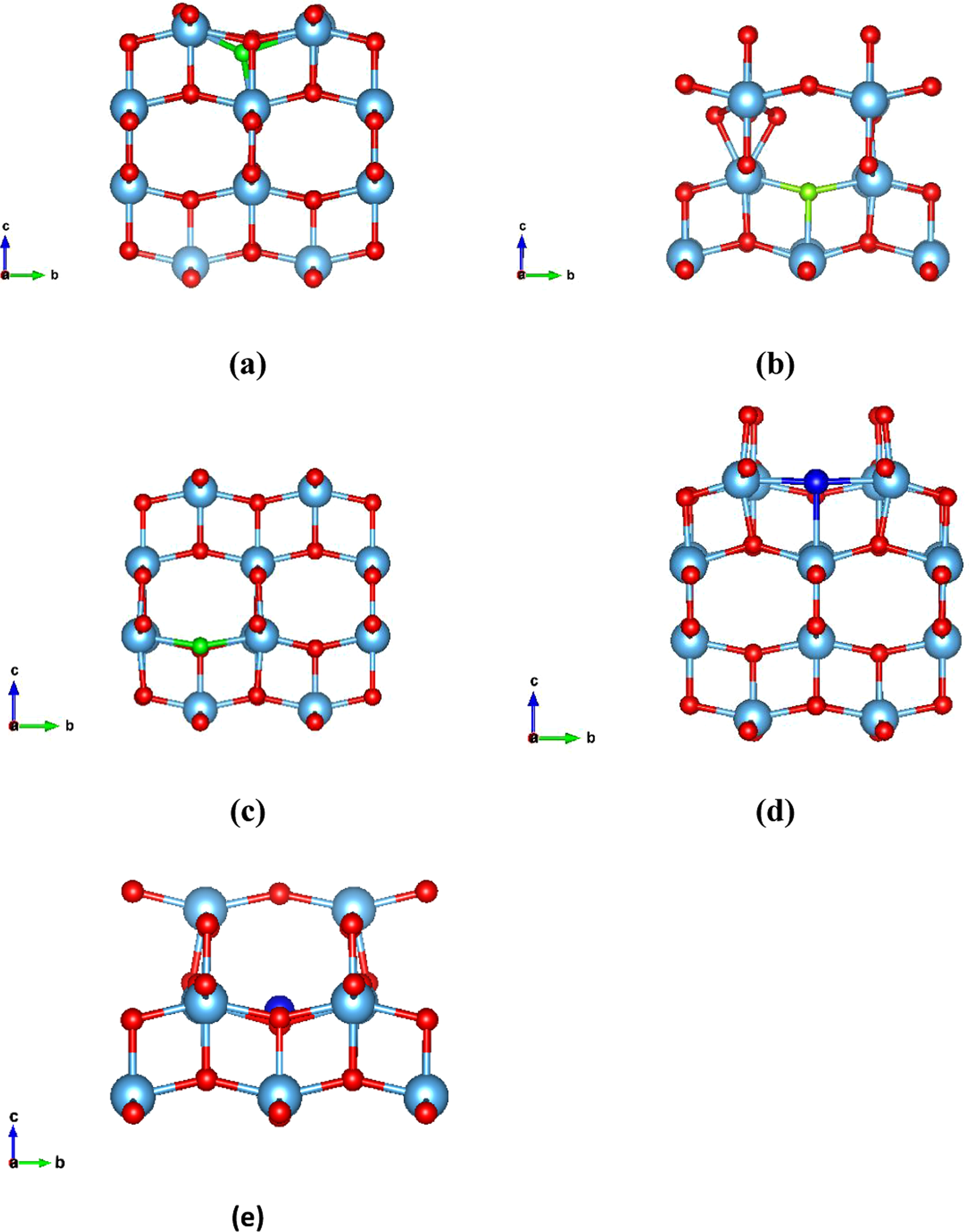
Although a profound band gap reduction can be beneficial to the material’s photocatalytic activity as it results in higher absorption of visible light, it might create mid gap states that usually act as charge traps hence having a negative impact on the performance of organic and perovskite solar cells utilizing TiO2 exclusively as electron transport/extraction material. In those cases the photocatalytic ability of TiO2 should be suppressed as it degrades its interface with organic/perovskite semiconductor. The formation of mid gap states upon doping of TiO2 is therefore undesired in OSCs and PSCs. In the present study, fluorine and chlorine doping of the bulk and surface TiO2 is studied via Density Functional Theory (DFT) in order to examine the electrical structure before and after doping with F and Cl and investigate the potential improvement in the photocatalytic activity of the TiO2. Moreover we investigated many different defect sites for the F and Cl in the bulk system and we also calculated for the first time the interstitial sites and the changes in electrical properties of the (001) TiO2 surface after the F and Cl doping. Total density of states (DOS) and partial DOS (PDOS) of the energetically minimum sites of the defects are considered in order for the electrical structure changes to be fully understood. A band gap reduction is evident in both cases. Moreover, the formation of mid gap states in all cases is also predicted. Such states are highly beneficial for the photocatalytic applications of TiO2 though they can be detrimental for OSCs and PSCs performance as they constitute trap sites for the photogenerated charge carriers thus significantly reducing the device photocurrent.
Get funded, develop & make a difference!